|
Sources, facts, uses, scarcity (SRI), podcasts, alchemical symbols, videos and images. Element Antimony (Sb), Group 15, Atomic Number 51, p-block, Mass 121.760. Noble gases are a set of six chemical elements in the periodic table that have. Periodic Table of Videos Created by video journalist Brady Haran working with chemists at The University of Nottingham. The most common reactive nonmetals are carbon, nitrogen, and oxygen. Nevertheless, even these 18 tend to become metallic at huge pressures. Podcasts Produced by The Naked Scientists. The number of electrons in the outermost shell of a particular atom determines its reactivity, or tendency to form chemical bonds with other atoms. 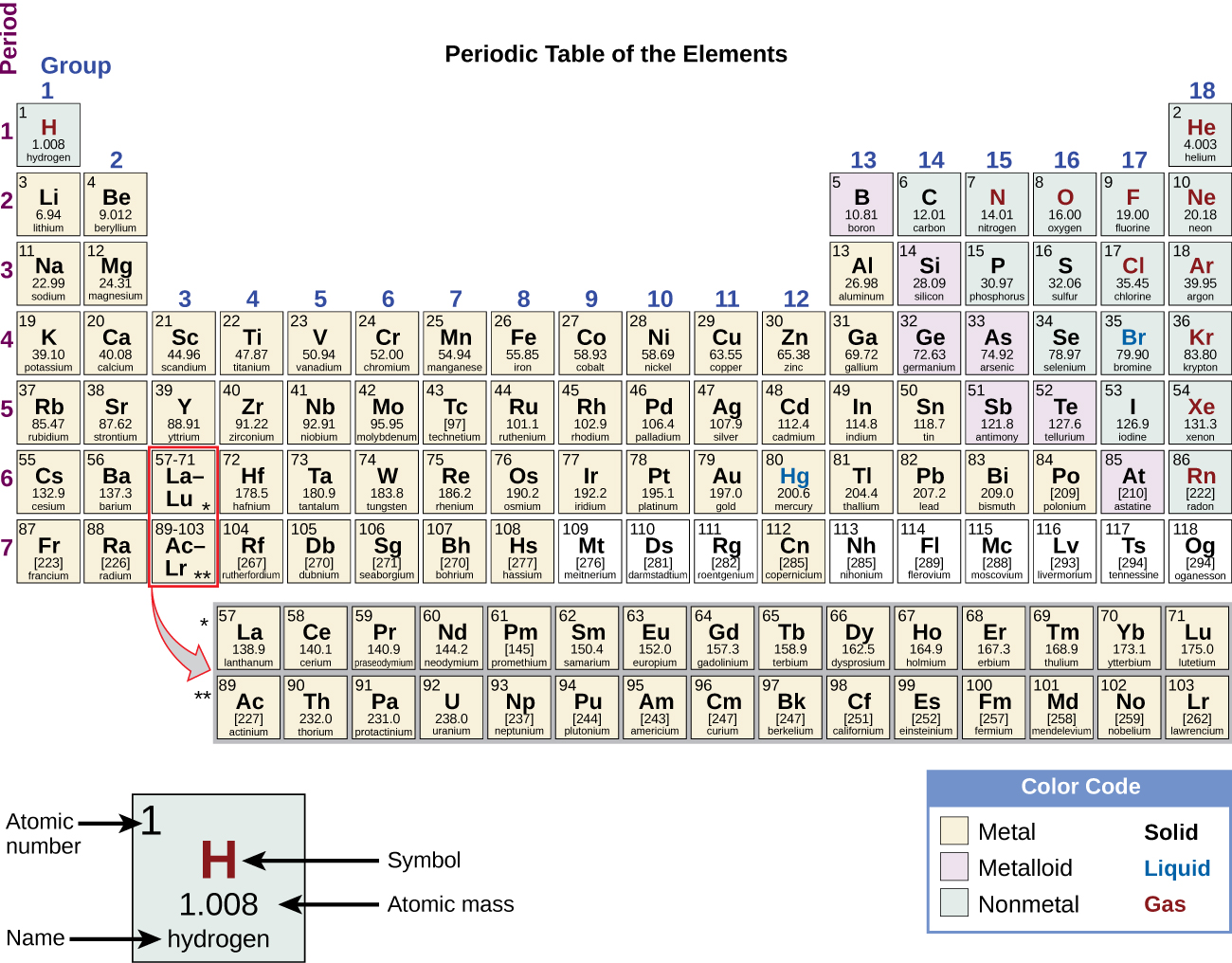
Many nonmetals (hydrogen, nitrogen, oxygen, fluorine, chlorine, bromine, and iodine) are diatomic, and most of the rest are polyatomic. Bulk tissues of living organisms are composed almost entirely of nonmetals. Only eighteen elements in the periodic table are generally considered nonmetals, compared to over eighty metals, but nonmetals make up most of the crust, atmosphere and oceans of the earth. nonmetals usually have little or no luster.they have significantly lower melting points and boiling points than metals.usually have lower densities than metals.Since the noble gases are a special group because of their lack of reactivity, the element fluorine is the most reactive nonmetal. The most reactive nonmetals reside in the upper right portion of the periodic table. Nonmetals will only ever form one negative ion. Main group metals - those in groups 1 and 2, plus aluminum, form just one positive ion. in solid form, they are dull and brittle, rather than metals which are lustrous, ductile or malleable Nonmetals tend to gain electrons in chemical reactions, and have a high attraction for electrons within a compound. The periodic table organizes elements into vertical groups.In group 1 of the periodic table, the first element is a nonmetal, the second and. they form acidic oxides (whereas metals generally form basic oxides) periodic table which was compared with the octaves of music failed.poor conductors of heat and electricity when compared to metals.Common properties considered characteristic of a nonmetal include: In 1870, the elements with a yellow foreground were known. One of the various types of Periodic Table designs is the ‘short form,’ an example being displayed in Figure 1. There is no rigorous definition for the term "nonmetal" - it covers a general spectrum of behaviour. The Periodic Rule is specific for the selected field of chemistry such as at ambient human (or planetary core, or cosmic space) conditions. All elements in Group 18 - the noble gases.All elements in Group 17 - the halogens.Several elements in Group 16, the chalcogens: oxygen (O), sulfur (S), selenium (Se).In Group 15 (the pnictogens): nitrogen (N), phosphorus (P) On the periodic table, non-metals lie to the right of the zigzag line that runs between the elements boron, silicon, arsenic, tellurium, and astatine.Solids, liquids or gases at room temperature.The elements generally regarded as nonmetals are:.Poor conductors of heat and electricity.Malleable - can be beaten into thin sheets.  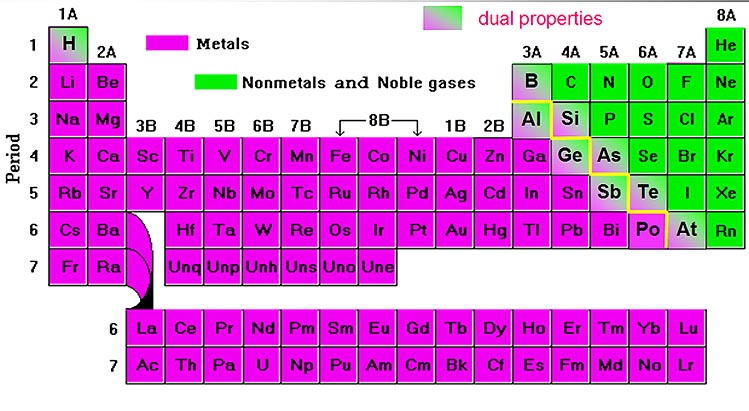
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
